Trung Tâm Luyện Thi Đại Học
Chapter 1. Chemical Reactions and Equations
1.2. Types Of Chemical Reactions
We have learnt in Class IX that during a chemical reaction atoms of one element do not change into those of another element. Nor do atoms disappear from the mixture or appear from elsewhere. Actually, chemical reactions involve the breaking and making of bonds between atoms to produce new substances. You will study about types of bonds formed between atoms in Chapters 3 and 4.
1.2.1 Combination Reaction
Activity 1.4
Take a small amount of calcium oxide or quick lime in a beaker.
- Slowly add water to this.
- Touch the beaker as shown in Fig. 1.3.
- Do you feel any change in temperature?
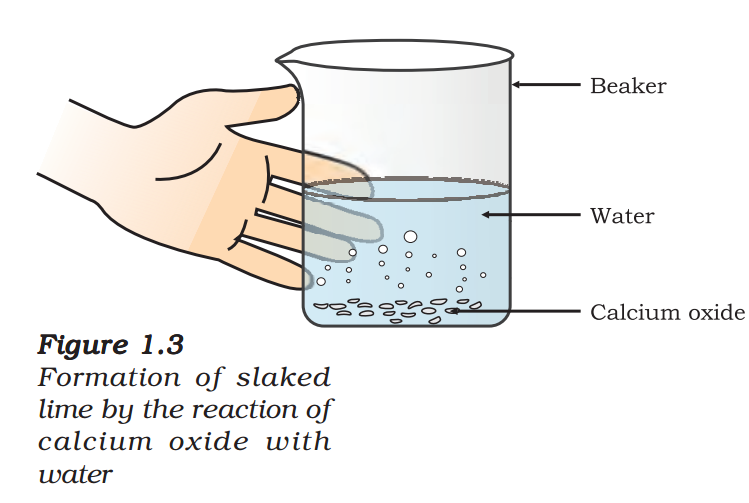
Calcium oxide reacts vigorously with water to produce slaked lime (calcium hydroxide) releasing a large amount of heat.
\( \begin{align} & CaO(s)+{{H}_{2}}O(\ell )\to Ca{{(OH)}_{2}}(aq)+Heat\,\,\,\,\,\,(1.13) \\ & (\text{Quick}\,\,\text{lime})\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(\text{Slaked}\,\,\text{lime}) \\ \end{align} \)
In this reaction, calcium oxide and water combine to form a single product, calcium hydroxide. Such a reaction in which a single product is formed from two or more reactants is known as a combination reaction.
Do you know?
A solution of slaked lime produced by the reaction 1.13 is used for whitewashing walls. Calcium hydroxide reacts slowly with the carbon dioxide in air to form a thin layer of calcium carbonate on the walls. Calcium carbonate is formed after two to three days of whitewashing and gives a shiny finish to the walls. It is interesting to note that the chemical formula for marble is also CaCO3.
\( \begin{align} & Ca{{(OH)}_{2}}(aq)+C{{O}_{2}}(g)\to CaC{{O}_{3}}(s)+{{H}_{2}}O(\ell )\,\,\,\,\,\,\,(1.14) \\& (Calcium\,\,hydroxide)\,\,\,\,\,\,\,(Calcium\,\,carbonate) \\ \end{align} \)
Let us discuss some more examples of combination reactions.
(i) Burning of coal
\( C(s)+{{O}_{2}}(g)\to C{{O}_{2}}(g)\,\,\,\,\,\,\,\,\,\,(1.15) \)
(ii) Formation of water from H2(g) and O2(g)
\( 2{{H}_{2}}(g)+{{O}_{2}}(g)\to 2{{H}_{2}}O(\ell )\,\,\,\,\,\,\,(1.16) \)
In simple language we can say that when two or more substances (elements or compounds) combine to form a single product, the reactions are called combination reactions.
In Activity 1.4, we also observed that a large amount of heat is evolved.
This makes the reaction mixture warm. Reactions in which heat is released along with the formation of products are called exothermic chemical reactions.
Other examples of exothermic reactions are –
(i) Burning of natural gas
\( C{{H}_{4}}(g)+2{{O}_{2}}(g)\to C{{O}_{2}}(g)+2{{H}_{2}}O\,\,\,\,\,\,\,\,\,(1.17) \)
(ii) Do you know that respiration is an exothermic process?
We all know that we need energy to stay alive. We get this energy from the food we eat. During digestion, food is broken down into simpler substances. For example, rice, potatoes and bread contain carbohydrates. These carbohydrates are broken down to form glucose.
This glucose combines with oxygen in the cells of our body and provides energy. The special name of this reaction is respiration, the process of which you will study in Chapter 6.
\( \begin{align} & {{C}_{6}}{{H}_{12}}{{O}_{6}}(aq)+6{{O}_{2}}(aq)\to 6C{{O}_{2}}(aq)+6{{H}_{2}}O(\ell )+energy\,\,\,\,\,(1.18) \\ & (\text{Glucose}) \\ \end{align} \)
(iii) The decomposition of vegetable matter into compost is also an example of an exothermic reaction.
Identify the type of the reaction taking place in Activity 1.1, where heat is given out along with the formation of a single product.
1.2.2 Decomposition Reaction
Activity 1.5
- Take about 2 g ferrous sulphate crystals in a dry boiling tube.
- Note the colour of the ferrous sulphate crystals.
- Heat the boiling tube over the flame of a burner or spirit lamp as shown in Fig. 1.4.
- Observe the colour of the crystals after heating.
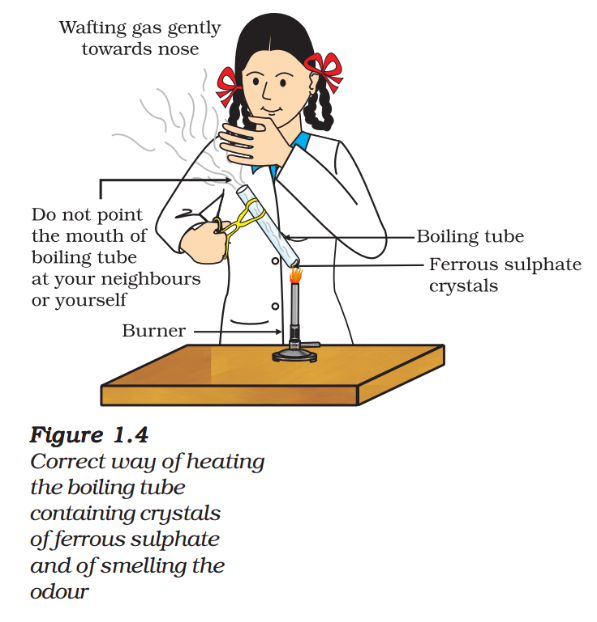
Have you noticed that the green colour of the ferrous sulphate crystals has changed? You can also smell the characteristic odour of burning sulphur.
\( \begin{align} &2FeS{{O}_{4}}(s)\xrightarrow{Heat}F{{e}_{2}}{{O}_{3}}(s)+S{{O}_{2}}(g)+S{{O}_{3}}(g)\,\,\,\,\,\,\,\,(1.19) \\ & (Ferrous\,\,sulphate)\,\,\,\,\,\,\,(Ferric\,\,oxide) \\ \end{align} \)
In this reaction you can observe that a single reactant breaks down to give simpler products. This is a decomposition reaction. Ferrous sulphate crystals (FeSO4, 7H2O) lose water when heated and the colour of the crystals changes. It then decomposes to ferric oxide (Fe2O3), sulphur dioxide (SO2) and sulphur trioxide (SO3). Ferric oxide is a solid, while SO2 and SO3 are gases.
Decomposition of calcium carbonate to calcium oxide and carbon dioxide on heating is an important decomposition reaction used in various industries. Calcium oxide is called lime or quick lime. It has
many uses – one is in the manufacture of cement. When a decomposition reaction is carried out by heating, it is called thermal decomposition.
\( \begin{align} & CaC{{O}_{3}}(s)\xrightarrow{Heat}CaO(s)+C{{O}_{2}}(g)\,\,\,\,\,\,\,(1.20) \\ & (Limestone)\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(\text{Quick}\,\,\text{lime}) \\ \end{align} \)
Another example of a thermal decomposition reaction is given in Activity 1.6.
Activity 1.6
- Take about 2 g lead nitrate powder in a boiling tube.
- Hold the boiling tube with a pair of tongs and heat it over a flame, as shown in Fig. 1.5.
- What do you observe? Note down the change, if any.
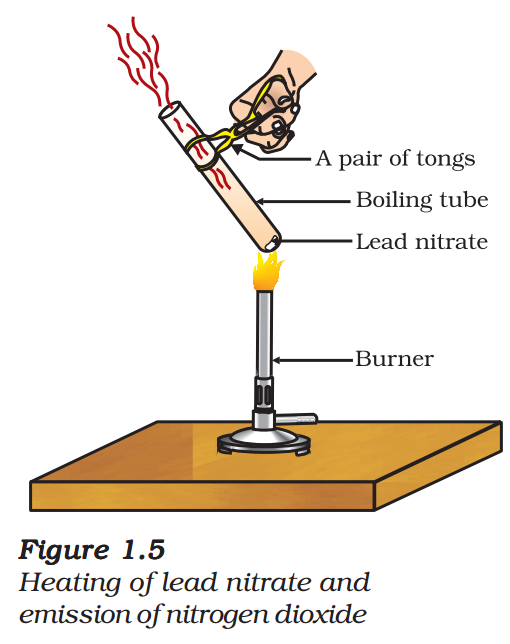
You will observe the emission of brown fumes. These fumes are of nitrogen dioxide (NO2). The
reaction that takes place is –
\( \begin{align} &2Pb{{(N{{O}_{3}})}_{2}}(s)\,\,\,\,\,\xrightarrow{Heat}2PbO(s)\,\,\,\,\,\,\,\,+\,\,\,\,\,\,2N{{O}_{2}}(g)\,\,\,\,\,\,\,\,\,\,\,\,\,+\,\,\,\,\,\,\,{{O}_{2}}(g)\,\,\,\,\,\,\,(1.21) \\ &(Lead\,\,nitrate)\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Lead\,\,oxide)\,\,\,\,\,\,\,(Nitrogen\,\,dioxide)\,\,\,\,\,\,(Oxygen) \\ \end{align} \)
Let us perform some more decomposition reactions as given in Activities 1.7 and 1.8.
Activity 1.7
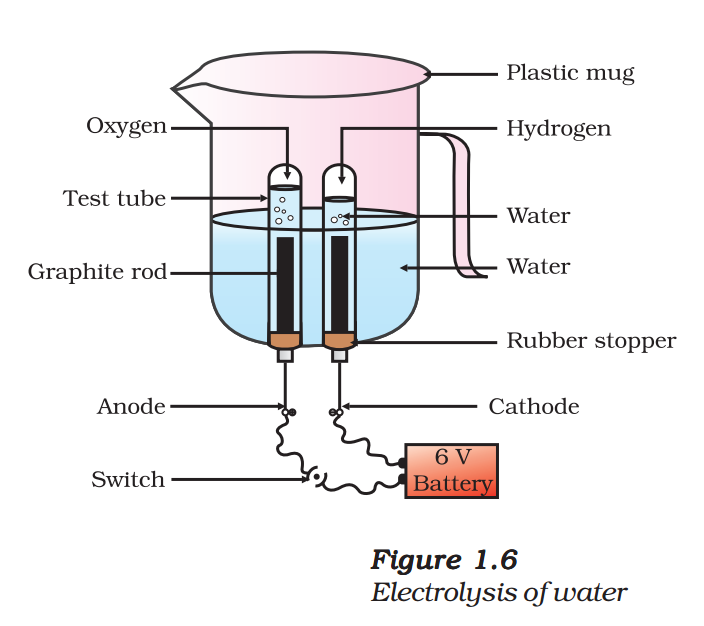
- Take a plastic mug. Drill two holes at its base and fit rubber stoppers in these holes.
Insert carbon electrodes in these rubber stoppers as shown in Fig. 1.6. - Connect these electrodes to a 6 volt battery.
- Fill the mug with water such that the electrodes are immersed. Add a few drops of dilute sulphuric acid to the water.
- Take two test tubes filled with water and invert them over the two carbon electrodes.
- Switch on the current and leave the apparatus undisturbed for some time.
- You will observe the formation of bubbles at both the electrodes. These bubbles displace water in the test tubes.
- Is the volume of the gas collected the same in both the test tubes?
- Once the test tubes are filled with the respective gases, remove them carefully.
Test these gases one by one by bringing a burning candle close to the mouth of the test tubes.
CAUTION: This step must be performed carefully by the teacher.
- What happens in each case?
- Which gas is present in each test tube?
Activity 1.8
- Take about 2 g silver chloride in a china dish.
- What is its colour?
- Place this china dish in sunlight for some time (Fig. 1.7).
- Observe the colour of the silver chloride after some time.
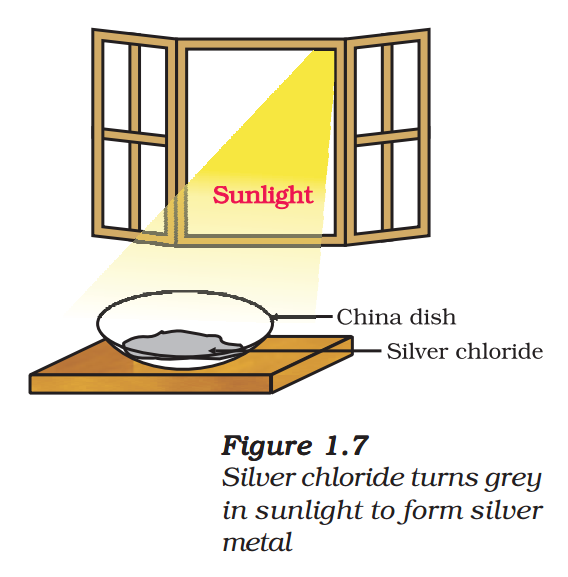
You will see that white silver chloride turns grey in sunlight. This is due to the decomposition of silver chloride into silver and chlorine by light.
\( 2AgCl(s)\xrightarrow{Sunlight}2Ag(s)+C{{l}_{2}}(g)\,\,\,\,\,\,\,\,\,(1.22) \)
Silver bromide also behaves in the same way.
\( 2AgBr(s)\xrightarrow{Sunlight}2Ag(s)+B{{r}_{2}}(s)\,\,\,\,\,\,\,(1.23) \)
The above reactions are used in black and white photography.
What form of energy is causing these decomposition reactions?
We have seen that the decomposition reactions require energy either in the form of heat, light or electricity for breaking down the reactants. Reactions in which energy is absorbed are known as endothermic reactions.
Carry out the following Activity
Take about 2 g barium hydroxide in a test tube. Add 1 g of ammonium chloride and mix with the help of a glass rod. Touch the bottom of the test tube with your palm. What do you feel? Is this an exothermic or endothermic reaction?
Questions
1. A solution of a substance ‘X’ is used for whitewashing.
(i) Name the substance ‘X’ and write its formula.
(ii) Write the reaction of the substance ‘X’ named in (i) above with water.
2. Why is the amount of gas collected in one of the test tubes in Activity 1.7 double of the amount collected in the other? Name this gas.
1.2.3 Displacement Reaction
Activity 1.9
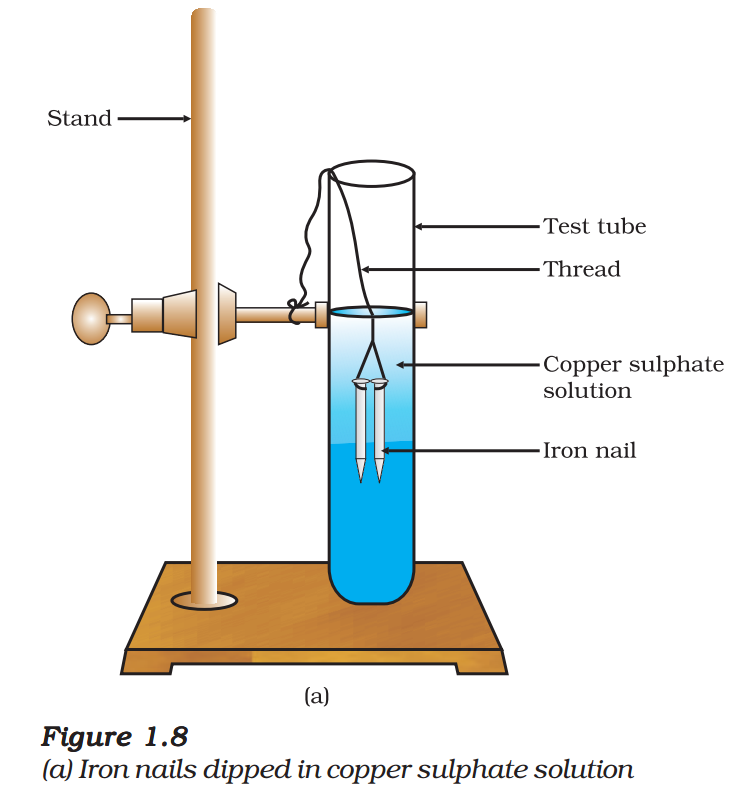
- Take three iron nails and clean them by rubbing with sand paper.
- Take two test tubes marked as (A) and (B). In each test tube, take about 10 mL copper sulphate solution.
- Tie two iron nails with a thread and immerse them carefully in the copper sulphate solution in test tube B for
about 20 minutes [Fig. 1.8 (a)]. Keep one iron nail aside for comparison. - After 20 minutes, take out the iron nails from the copper sulphate solution.
- Compare the intensity of the blue colour of copper sulphate solutions in test tubes (A) and (B) [Fig. 1.8 (b)].
- Also, compare the colour of the iron nails dipped in the copper sulphate solution with the one kept aside [Fig. 1.8 (b)].
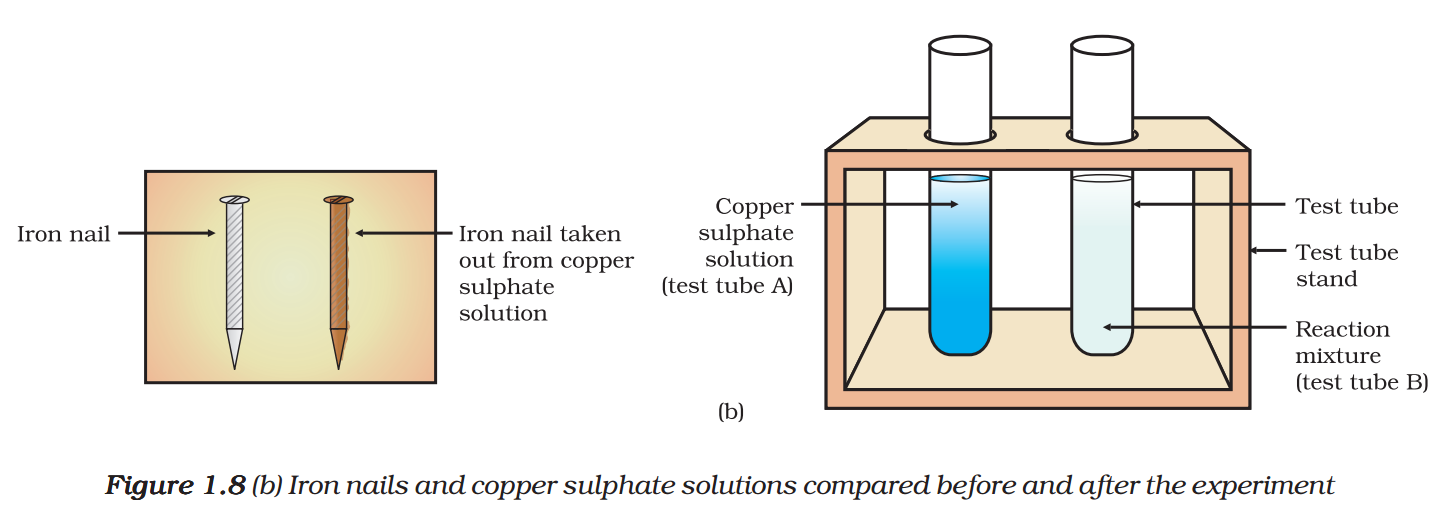
Why does the iron nail become brownish in colour and the blue colour of copper sulphate solution fades?
The following chemical reaction takes place in this Activity–
\(\begin{align} & Fe(s)+CuS{{O}_{4}}(aq)\,\,\,\,\,\,\,\,\,\,\to\,\,\,\,\,\,FeS{{O}_{4}}(aq)+Cu(s)\,\,\,\,\,\,\,\,(1.24) \\ & \,\,\,\,\,\,\,\,\,\,\,(Copper\,\,sulphate)\,\,\,\,\,\,(Iron\,\,sulphate) \\ \end{align}\)
In this reaction, iron has displaced or removed another element, copper, from copper sulphate solution. This reaction is known as displacement reaction.
Other examples of displacement reactions are
\( \begin{align} & Zn(s)+CuS{{O}_{4}}(aq)\,\,\,\,\,\,\,\,\,\to\,\,\,\,\,\,ZnS{{O}_{4}}(aq)+Cu(s)\,\,\,\,\,(1.25) \\ &\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Copper\,\,sulphate)\,\,\,\,\,(Zinc\,\,sulphate) \\ \end{align} \)
\( \begin{align} & Pb(s)+CuC{{l}_{2}}(aq)\,\,\,\,\,\,\to\,\,\,\,\,\,PbC{{l}_{2}}(aq)+Cu(s)\,\,\,\,\,\,\,\,\,(1.26) \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,(Copper\,\,chloride)\,\,\,\,(Lead\,\,chloride) \\ \end{align} \)
Zinc and lead are more reactive elements than copper. They displace copper from its compounds.
1.2.4 Double Displacement Reaction
Activity 1.10
- Take about 3 mL of sodium sulphate solution in a test tube.
- In another test tube, take about 3 mL of barium chloride solution.
- Mix the two solutions (Fig. 1.9).
- What do you observe?
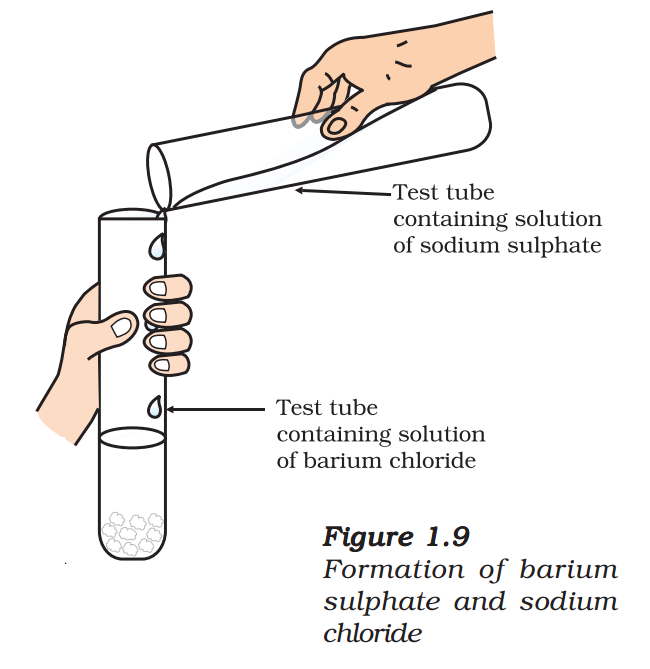
You will observe that a white substance, which is insoluble in water, is formed. This insoluble substance formed is known as a precipitate. Any reaction that produces a precipitate can be called a precipitation reaction.
\( \begin{align} & N{{a}_{2}}S{{O}_{4}}(aq)\,\,\,\,\,\,\,\,\,\,+\,\,\,\,\,\,\,BaC{{l}_{2}}(aq)\,\,\,\,\,\,\,\,\,\to\,\,\,\,\,\,\,BaS{{O}_{4}}(s)\,\,\,\,\,\,\,\,\,\,+\,\,\,\,\,\,\,2NaCl(aq)\,\,\,\,\,\,\,\,\,\,(1.27) \\ &(Sodium\,\,sulphate)\,\,\,\,\,\,(Barium\,\,chloride)\,\,\,\,\,(Barium\,\,sulphate)\,\,\,(Sodium\,\,chloride) \\ \end{align} \)
What causes this? The white precipitate of BaSO4 is formed by the reaction of \( SO_{4}^{2-} \) and Ba2+. The other product formed is sodium chloride which remains in the solution. Such reactions in which there is an
exchange of ions between the reactants are called double displacement reactions.
Recall Activity 1.2.
Where you have mixed the solutions of lead(II) nitrate and potassium iodide.
(i) What was the colour of the precipitate formed? Can you name the compound precipitated?
(ii) Write the balanced chemical equation for this reaction.
(iii) Is this also a double displacement reaction?
1.2.5 Oxidation and Reduction
Activity 1.11
- Heat a china dish containing about 1 g copper powder (Fig. 1.10).
- What do you observe?
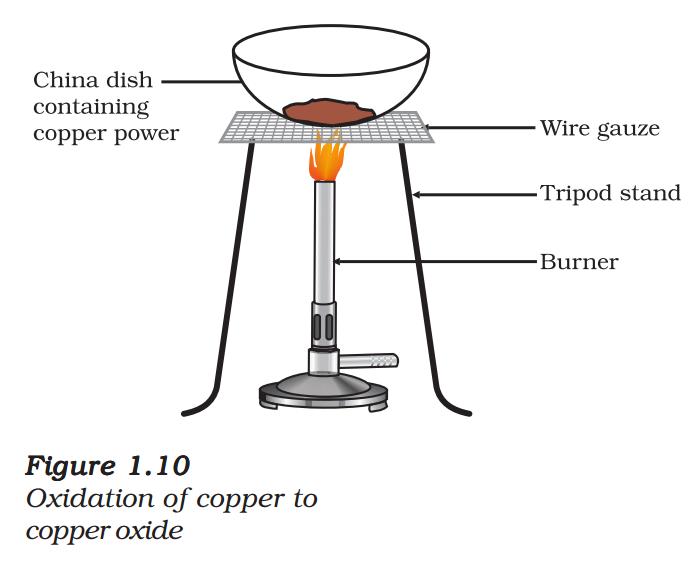
The surface of copper powder becomes coated with black copper(II) oxide. Why has this black substance formed?
This is because oxygen is added to copper and copper oxide is formed.
\( 2Cu+{{O}_{2}}\xrightarrow{Heat}2CuO\,\,\,\,\,\,\,\,\,\,(1.28) \)
If hydrogen gas is passed over this heated material (CuO), the black coating on the surface turns brown as the reverse reaction takes place and copper is obtained.
\( CuO+{{H}_{2}}\xrightarrow{Heat}Cu+{{H}_{2}}O\,\,\,\,\,\,\,\,\,\,(1.29) \)
If a substance gains oxygen during a reaction, it is said to be oxidised.
If a substance loses oxygen during a reaction, it is said to be reduced.
During this reaction (1.29), the copper(II) oxide is losing oxygen and is being reduced. The hydrogen is gaining oxygen and is being oxidised.
In other words, one reactant gets oxidised while the other gets reduced during a reaction. Such reactions are called oxidation-reduction reactions or redox reactions.

Some other examples of redox reactions are:
\( ZnO+C\to CO\,\,\,\,\,\,\,\,\,\,(1.31) \)
\( Mn{{O}_{2}}+4HCl\to MnC{{l}_{2}}+2{{H}_{2}}O+C{{l}_{2}}\,\,\,\,\,\,\,\,\,\,(1.32) \)
In reaction (1.31) carbon is oxidised to CO and ZnO is reduced to Zn.
In reaction (1.32) HCl is oxidised to Cl2 whereas MnO2 is reduced to MnCl2.
From the above examples we can say that if a substance gains oxygen or loses hydrogen during a reaction, it is oxidised. If a substance loses oxygen or gains hydrogen during a reaction, it is reduced.
Recall Activity 1.1
Where a magnesium ribbon burns with a dazzling flame in air (oxygen) and changes into a white substance, magnesium oxide. Is magnesium being oxidised or reduced in this reaction?
Các bài toán cùng chủ đề!
Các sách luyện thi do Trung tâm phát hành!
Trung Tâm Luyện Thi Đại Học được xây dựng trên WordPress





